
Modern surgical environments operate under competing priorities that are always in play. Clinical teams need dependable and consistent instrument performance and verified sterility. Supply chain leaders are tasked with stabilizing costs amid fluctuating demand. Finance departments look for predictability, while sustainability teams increasingly ask harder questions about waste and environmental impact. Yet the information required to make confident decisions about instrumentation often lives in separate systems, departments, or assumptions rather than shared evidence.
Hospitals today are not simply choosing between reusable and disposable tools. They are evaluating reliability, workflow stability, and long-term operational resilience. When conversations lack structure, decisions tend to rely on price comparisons or anecdotal experience instead of measurable performance.
This guide offers a practical framework for evaluating Single-Use Surgery-Ready™ Surgical Instruments through the lenses that matter most in real surgical settings. Rather than presenting theory, the goal is to clarify what to measure, what questions to ask, and how organizations can make balanced decisions that stand up to daily operating room demands.
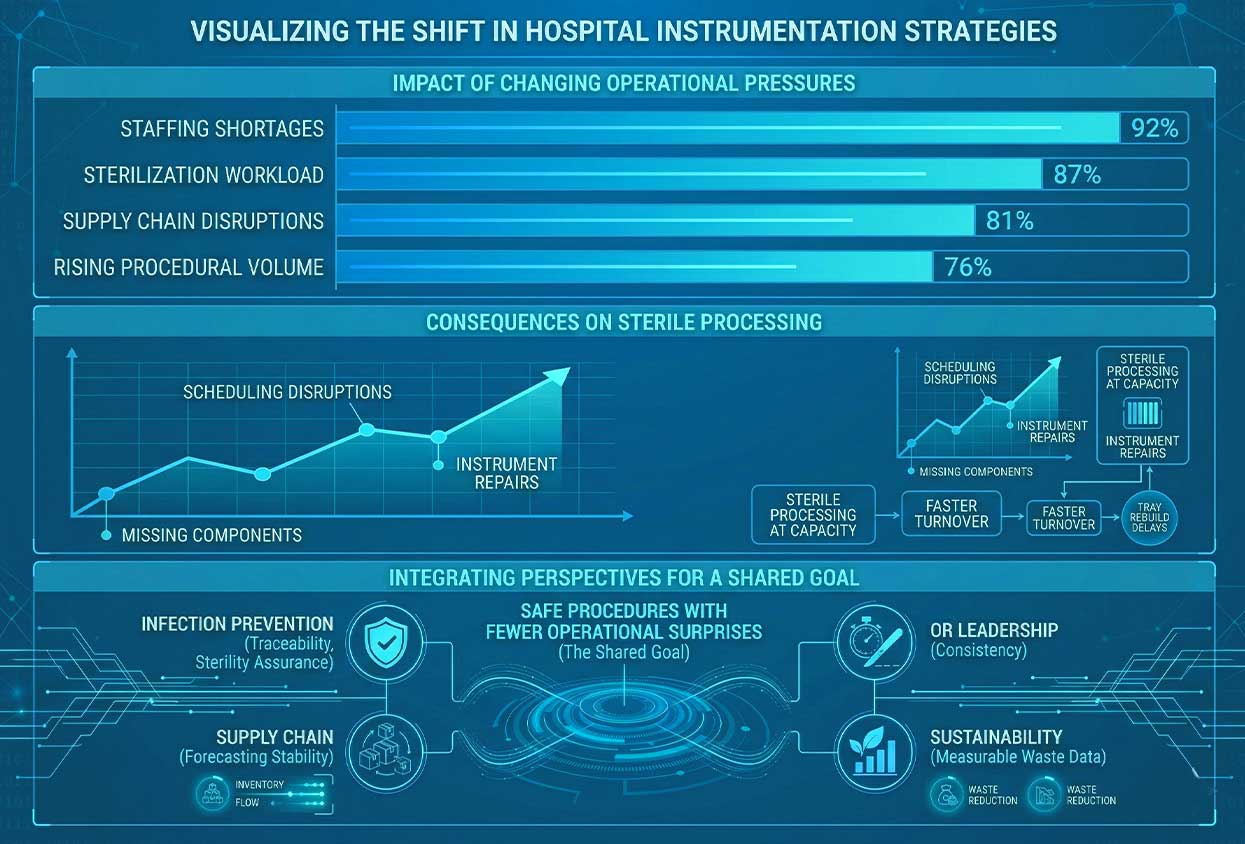
Healthcare systems are reassessing instrumentation strategies not because of trends but because operational pressures have changed. Staffing shortages, rising procedural volume, sterilization workload, and supply disruptions have reshaped how hospitals think about readiness.
Sterile processing departments frequently operate near capacity. Faster turnovers leave less margin for tray rebuild delays. Instrument repairs or missing components can cascade into scheduling disruptions that affect entire surgical days. These realities influence purchasing decisions just as much as clinical preference.
Within this environment, Single-Use Surgical Instruments are increasingly evaluated as part of broader workflow planning rather than isolated product substitutions. Decision-making now reflects shared institutional goals. Operating room leadership prioritizes consistency. Infection prevention teams focus on traceability and sterility assurance. Supply chain departments seek forecasting stability, while sustainability leaders want measurable waste data.
A good assessment aligns these perspectives instead of favoring one over another. The common objective remains simple: safe procedures with fewer operational surprises.
Safety discussions often become clouded by marketing language. In practice, safety means consistency. Instruments should function predictably, packaging should remain intact through storage and transport, and labeling must clearly support correct use every time.
Operational sterility refers not only to validated sterilization processes but also to maintaining package integrity until the moment of use. A sterile device that experiences compromised handling loses its intended value regardless of manufacturing quality.
Evaluating Single-Use Surgical-Ready™ Surgical Instruments begins with a documentation review. Instructions for use should be clear and accessible. Validation summaries, where applicable, demonstrate how sterility and performance standards are verified. Unique device identification labeling supports tracking and accountability, while defined change control processes confirm that updates are communicated transparently.
Observation inside the operating room provides equally valuable insight. Teams should evaluate ease of handling, tactile feedback, visibility within the surgical field, and whether instruments support consistent technique across users. Receiving and storage practices also deserve attention. Durable packaging, shelf-life controls, and realistic handling requirements help prevent unexpected failures before instruments ever reach the surgical suite.
Precision becomes especially important in fixation procedures. Torque-limiting instruments, for example, are designed to help apply intended tightening forces more consistently. These tools support procedural discipline by reducing variation during implant fixation workflows. While no instrument guarantees outcomes, consistent mechanical performance contributes to reliable execution.
One of the most common assessment errors occurs when organizations compare only the acquisition price. Instrument cost extends far beyond the invoice.
Reusable systems require labor for cleaning, inspection, assembly, sterilization, and transport. Facilities absorb expenses related to reprocessing supplies, equipment maintenance, tray repair, and replacement cycles. Overtime costs may emerge when sterile processing workloads exceed staffing capacity. Case delays caused by missing or damaged instruments also introduce hidden financial consequences.
When evaluating Single-Use Surgical Instruments, cost per case becomes a more meaningful metric. Direct expenses include purchase price, shipping logistics, storage requirements, and expiration management. Operational considerations involve sterile processing labor shifts, reduced maintenance demands, and elimination of repair cycles associated with reusable devices.
Clinical workflow impacts must also be considered. Even small setup interruptions can disrupt tightly scheduled operating rooms. Delays tied to instrument readiness affect staffing utilization and room efficiency.
Decision-making becomes clearer when viewed through realistic scenarios. High-volume procedures with standardized workflows often benefit from predictable instrumentation models that simplify preparation. Conversely, procedures with significant variability may benefit from hybrid approaches that combine reusable specialty tools with single-use components where consistency is most valuable.
Waste discussions surrounding surgical instrumentation often oversimplify complex systems. Meaningful comparison requires defining waste carefully.
Healthcare facilities generate multiple waste streams, including regulated medical waste and standard disposal categories. Classification varies by institutional policy, meaning identical products may be processed differently between hospitals. Packaging necessary to maintain sterility must also be distinguished from excess material that can potentially be optimized.
Evaluating Single-Use Surgical Instruments responsibly requires acknowledging both sides of the equation. Reusable instrumentation consumes water, energy, detergents, and chemical disinfectants throughout repeated reprocessing cycles. These environmental inputs are frequently overlooked in sustainability discussions.
Single-use systems, by contrast, contribute to packaging volume and disposal considerations after use. The most accurate comparison occurs at the facility level, accounting for local waste contracts, sterilization infrastructure, and procedural mix.
Beyond measurement, workflow aesthetics subtly influence performance. Surgical environments filled with stacked trays and partially opened supplies create visual complexity that demands constant decision-making. A more streamlined setup often promotes calmer preparation rhythms. This visual clarity reflects operational discipline rather than marketing appeal.
Infection prevention programs rely heavily on process reliability. Each reprocessing cycle introduces opportunities for delay, rework, or compliance strain when workloads increase.
Reducing reliance on repeated reprocessing can simplify operational risk management. Because Single-Use Surgery-Ready™ Surgical Instruments arrive sterile and ready for use, certain workflow steps shift away from internal processing demands. This does not replace infection prevention protocols but may support compliance by reducing bottlenecks that challenge consistency.
Facilities evaluating suppliers should examine sterility assurance methods and packaging integrity testing. Traceability systems enable rapid identification of product lots during audits or investigations. Equally important is change control communication. Healthcare teams must understand how product updates are documented and implemented over time to maintain confidence in long-term use.
These practices reinforce accountability across departments while supporting dependable surgical readiness.
Inventory variability remains a persistent challenge for hospitals managing thousands of individual components across service lines. Missing instruments or unexpected shortages disrupt scheduling and increase stress across teams.
Procedure-aligned supplies help stabilize planning. When inventory consumption aligns closely with surgical volume, forecasting improves. Single-Use Surgical Instruments can support this predictability by linking inventory directly to case demand rather than tray availability.
Effective inventory evaluation includes rationalizing stock-keeping units where possible while maintaining flexibility for specialized procedures. Forecasting models benefit from usage patterns tied to procedure mix, allowing purchasing teams to anticipate demand more accurately.
Shelf life planning also plays a role. Proper storage conditions, expiration monitoring, and manageable storage footprints reduce waste while preserving readiness. When inventory signals become clearer, supply chain teams spend less time reacting to shortages and more time optimizing performance.
Clinical acceptance ultimately determines success. Surgeons must feel confident that instrumentation supports established techniques without compromising ergonomics or control.
Assessment of Single-Use Surgical Instruments should include evaluation of handling consistency and functional familiarity. Quality systems behind the product matter equally. Organizations benefit from suppliers operating within mature quality management frameworks that emphasize validation practices, traceability, and structured complaint resolution.
Logistical reliability influences long-term adoption. Facilities should review lead times, distribution stability, packaging durability, and availability across geographic regions. Program support also matters. Providers capable of incorporating feedback and maintaining configuration control enable healthcare systems to refine solutions as needs evolve.
Questions directed toward vendors often reveal organizational maturity. Understanding how suppliers transition from concept to fulfillment, maintain consistency across production lots, and incorporate post-market feedback helps decision makers gauge partnership readiness.
Thoughtful adoption begins with controlled evaluation rather than broad implementation. Hospitals often select a single procedure with a stable technique and predictable volume to initiate a pilot.
Stakeholder alignment remains essential. Operating room teams, sterile processing leadership, infection prevention specialists, procurement professionals, and sustainability representatives should agree on success criteria before implementation begins. Training should address receiving procedures, storage practices, opening technique, and documentation capture to ensure consistent evaluation conditions.
During the pilot, organizations assessing Single-Use Surgical Instruments typically monitor safety signals such as packaging integrity, labeling clarity, usability feedback, and any performance concerns observed during procedures. Operational observations may include setup consistency, reduction in missing instruments, and shifts in sterile processing workload.
Financial evaluation focuses on cost per case using the full operational model rather than acquisition price alone. Waste assessment considers disposal volume estimates alongside packaging observations to inform sustainability discussions.
Common mistakes include expanding programs before pilot data stabilizes, overlooking surgeon preferences early in the process, underestimating storage requirements, or treating sustainability goals as abstract commitments rather than measurable outcomes. A disciplined pilot structure builds confidence across departments and supports informed scaling.
Healthcare leaders face increasing pressure to balance safety, operational efficiency, financial responsibility, and sustainability goals simultaneously. A structured framework allows organizations to evaluate Single-Use Surgical Instruments with clarity rather than assumptions.
When safety checkpoints are defined, cost models reflect real operations, and waste discussions rely on measurable data, decision-making becomes more confident. The result is not simply a purchasing choice but a workflow improvement grounded in evidence.
Well-organized data brings a sense of calm to complex environments. Surgical teams work more confidently when instrumentation is predictable, inventory signals are clear, and pilots are measured carefully before expansion.
Healthcare organizations interested in exploring surgery-ready solutions can connect with ECA Medical to review procedural needs, develop a practical pilot scorecard, and evaluate options that support safety, inventory control, and consistent case readiness. ECA Medical continues to develop new kits for additional procedures, and facilities are encouraged to check back regularly or collaborate directly to design configurations tailored to evolving surgical demands.
Are Single-Use Surgical Instruments as safe as reusable instruments?
Safety depends on consistent manufacturing quality, verified sterility assurance, and proper clinical fit. Many facilities evaluate performance through structured pilots before broader adoption.
How can hospitals compare true cost per case fairly?
Include acquisition costs alongside labor, reprocessing resources, maintenance, delays, and workflow impacts to capture the full financial picture.
What documentation supports traceability?
Facilities typically request instructions for use, labeling with device identification, sterility assurance information, and defined change control communication.
How should waste comparisons be evaluated?
Measure both disposal volume and reprocessing resource consumption within the context of each facility’s infrastructure.
What pilot approach builds staff confidence?
Start with one procedure, align stakeholders early, collect measurable data, and expand only after consistent performance is demonstrated. Contact ECA Medical for more information today.